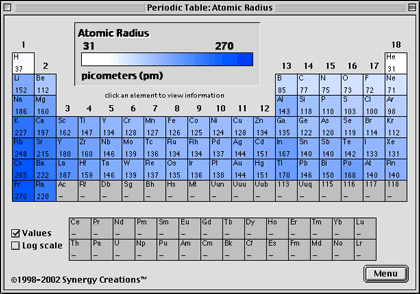
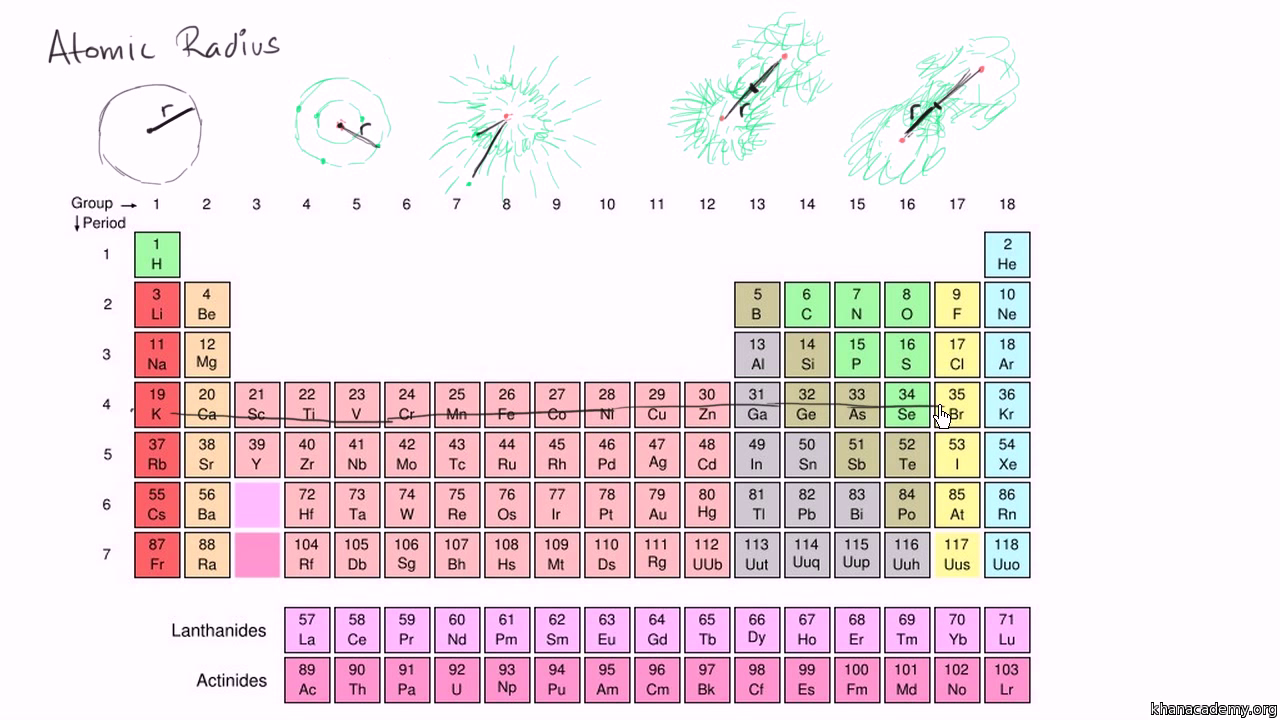
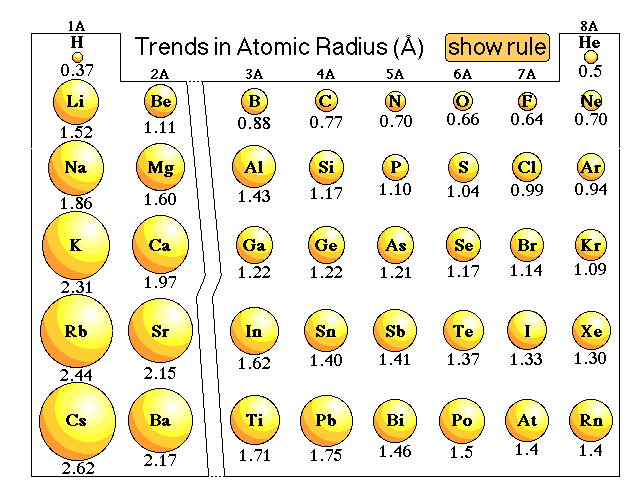
 In addition, the size of an atom’s electron cloud also affects its chemical properties.Ītoms with larger electron clouds are more likely to form covalent bonds, while atoms with smaller electron clouds are more likely to form ionic bonds.Īs a result, the size of an atom plays a significant role in determining its chemical properties.Ĭonsequently, very little is known about the element. Most notably, the size of an atom’s nucleus determines the types of elements it can form.įor example, atoms with large nuclei are more likely to form stable noble gases, while atoms with smaller nuclei are more likely to form reactive metals. The size of an atom influences its chemical properties in a variety of ways. How does the size of an atom affect its chemical properties?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
